|
In addition, the charge to mass ratio of ionized particles was found to be different and depended on the type of gasses. It is because the canal rays consist of positive ions formed as a result of gas ionization. The canal rays depend on the nature or type of gas present inside the glass tube and are different from cathode rays. The assembly of the cathode, intensity grid, focus grid, and accelerating anode (positive electrode) is called an electron gun. Some experiments were made with a tube similar to that shown in fig. The cathode ray is a beam of electrons which are emitted by the heated cathode (negative electrode) and accelerated toward the fluorescent screen. In addition, the canal rays are considered to be the nucleus of the gas which has differentiated properties compared to cathode rays. When the cathode rays (whose path was traced by the phosphorescence on the glass). When a high external voltage source is applied to the glass tube via electrodes, it ionizes the gas present inside the tube where the positive ions constitute the origin of the canal ray. Thomson used cathode rays to find the wave-particle duality of light. The rays which were moving in opposite to the cathode rays were called canal rays. Cathode rays are streams of electrons that are observed in vacuum tubes.A magnetic field bends the cathode rays into a closed circle. Michael Faraday demonstrated in 1830 that when electricity is transmitted through. This experiment shows that a cathode ray is made up of particles with a constant ratio of charge to mass ( e/m ). The modification is needed because the cathode where the electron beam originates is situated a distance a 3.2 0.5. Increase the external voltage source to several thousand volts, which results in the emission of faint luminous rays emerging from the cathode electrode holes. Cathode Ray Experiment was conducted by J.J Thomson to discover electrons.This leads to the completion of the circuit and apparatus setup. High voltage passing through the electrode ionizes the air and makes it conduct the electricity like a conductor.Connect the two electrodes (anode and cathode) to a high external voltage source.To maintain a low pressure inside the glass tube, evacuate the air through the vacuum pump.The glass tube pressure is minimized through vacuum pumps or evacuating the air.A high voltage source (external) is connected to the metal electrodes.A Cathode ray tube made of glass with two metal electrodes ( anode and cathode) on each differentiated side.However, such tubes must not be used with too high a voltage because if they are the electrons on impact with the glass / fluorescent screen would produce a potential health hazard - X-rays. Modern cathode ray tubes use a hot cathode source whereas the older types use a cold cathode which generally require higher voltages to operate. y 1 2 a y t 2 From the capacitor voltage U A and the distance of the capacitor plates d the electric force F of the field can be determined. So there is a compromise between the values of voltage and pressure. Having a high voltage across the tube means that an electron can gain more kinetic energy between collisions with the air molecules and thus a smaller distance between gas molecules (not quite so low a pressure) can be used. This means that the gas pressure must not to be too low thus decreasing the distance the electrons travel between collisions with the gas molecules. On the other hand if there are too few gas molecules the intensity of the emitted would be too small to observe the emitted light. So a low pressure ensures that the distance between collision is sufficiently large to enable the electron to cause excitation/ionisation of the gas molecules. If the cathode ray experiment uses a discharge tube which emits light then the electrons must be accelerated enough (obtain enough kinetic energy) between collision with the gas molecules in the tube to excite/ionise the gas molecules on collision with them. 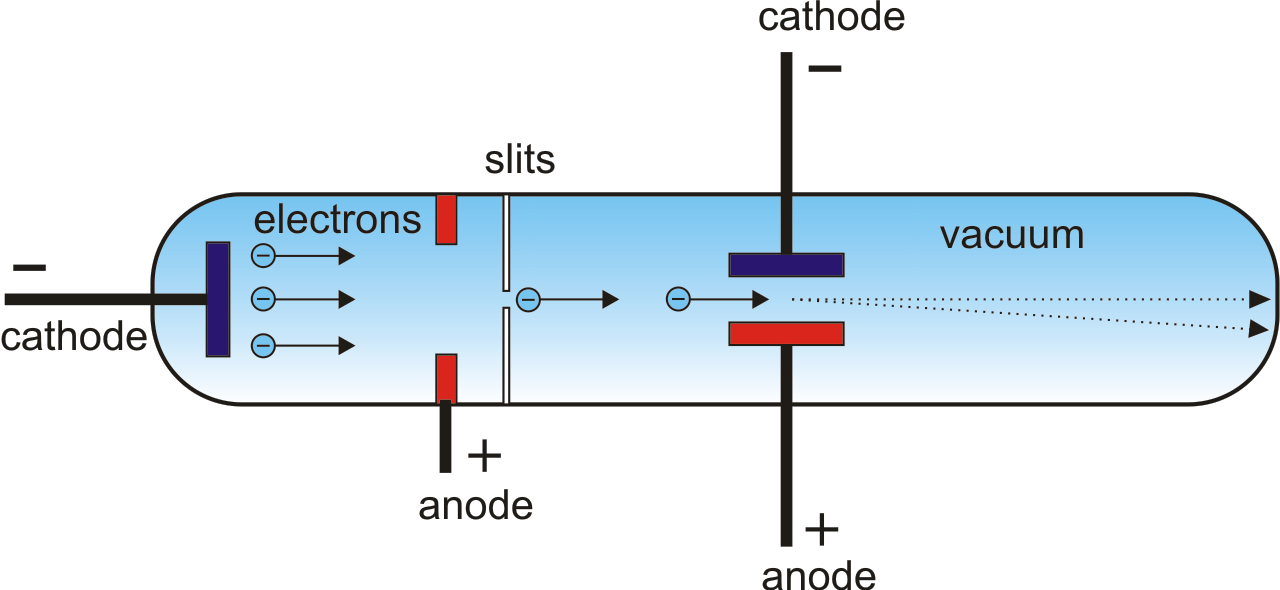 
You need a reasonably high accelerating voltage to give the electronsĮnough kinetic energy to make the fluorescent screen glow when the electrons hit the screen.You can also use the apparatus to demonstrate a valve diode a device that lets the current flow in just one direction. Of electrons is not impeded very much by collision with air molecules. Demonstration The main purpose of this experiment is to explain the principle of an electron gun. The tubes are called cathode ray tubes because the particle beam or 'cathode ray' originates at the cathode. You need the low pressure in the tube so that the passage of the beam A high voltage is applied across two electrodes at one end of the tube, which causes a beam of particles to flow from the cathode (the negatively-charged electrode) to the anode (the positively-charged electrode).  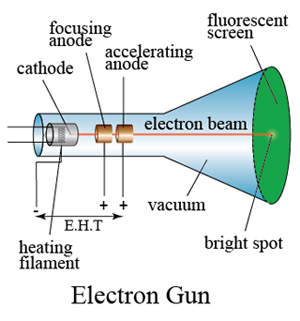
If the cathode ray experiment has a beam of electrons hitting a fluorescent screen which glows then:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
